
End to End Management of Clinical Trial Imaging
We provide all services necessary to support the Clinical Imaging Management system including training of site personnel, equipping the site with proper tools, quality assurance of the captured images, quality check of the acquired images, expert reading, adjudication, formatted reporting, till the submission of EDC.
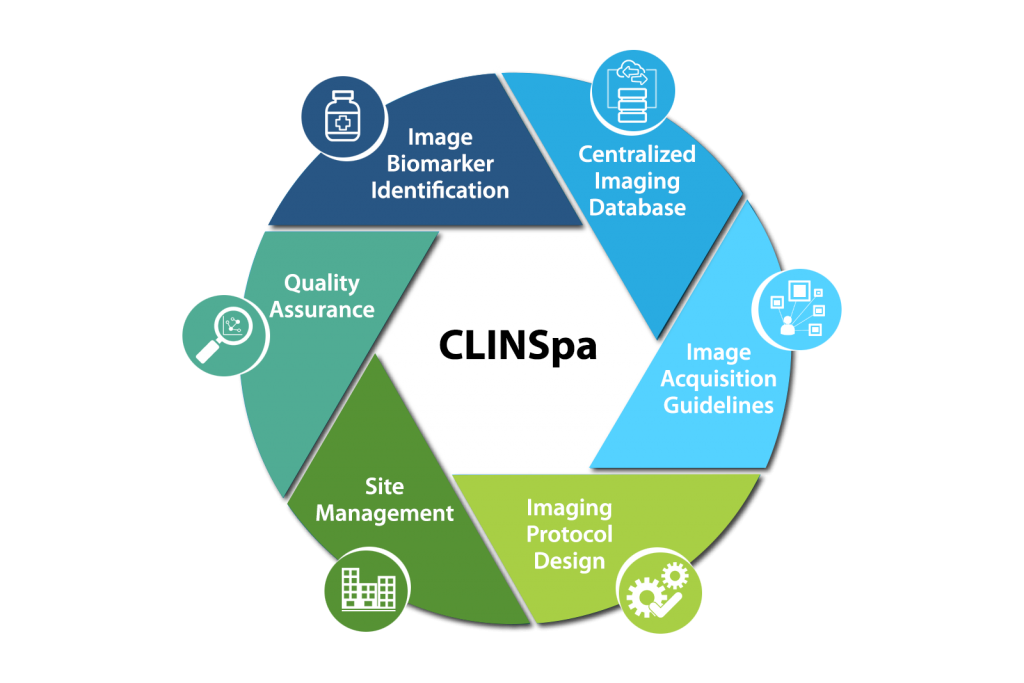
We accomplish this through a highly collaborative and integrated workflow based platform - CLINSpa.

Site Co-ordination, Site Management and Site Training
Clinical trials are incredibly complex and complexity increases when it comes to multi site trials with the huge enrollment of patients. Regardless of the study product or therapy area, clinical trial that needs imaging adds up to another layer of complexity, be it a regulatory challenge, workflow compliance challenge or training the site staff for proper image requirements.
So what should be done when one knows that equipping sites with proper tools and software is just not enough?
A successful imaging interpretation starts with proper training of the clinicians who are trained for the right image capturing technique. Successful site training regularizes imaging techniques that bring out quality image through simplified and consistent approaches.
Imaging Biomarker Identification
Biomarkers have become increasingly important to physicians, medical product developers (both drugs and devices), and regulatory agencies. As the incorporation of a biomarker into a clinical trial is becoming increasingly common, having a partner who can fully identify this component of the trial with experience and knowledge is critically important. ICL offers a full range of solutions for identifying the biomarker component of a clinical trial.
Radiology Imaging Protocol Design
On demand imaging protocol designing, done by a highly efficient panel of radiologists. So, whether you need modifications in your existing protocol or want it de Novo, we have experts who can cater to all your needs.

Quality Assurance
We utilize the latest advances in Imaging Interpretation and technology to ensure the best quality of image uploads and reads. We also digitize the medical images , develop and co - ordinate the imaging procedure in compliance with the FDA or other regulatory requirement.
Image Acquisition Guidelines
Do not settle for images that are distorted, blurred or has image projection errors such as elongation, shortening etc. Our Image Acquisition guidelines ensure standardized image capture throughout your trial period. Obtain and retrieve all your trial images effortlessly from multiple sites. Customized guidelines that suit every imaging need of your trial and brings out every minute details and findings that a radiologist may overlook.
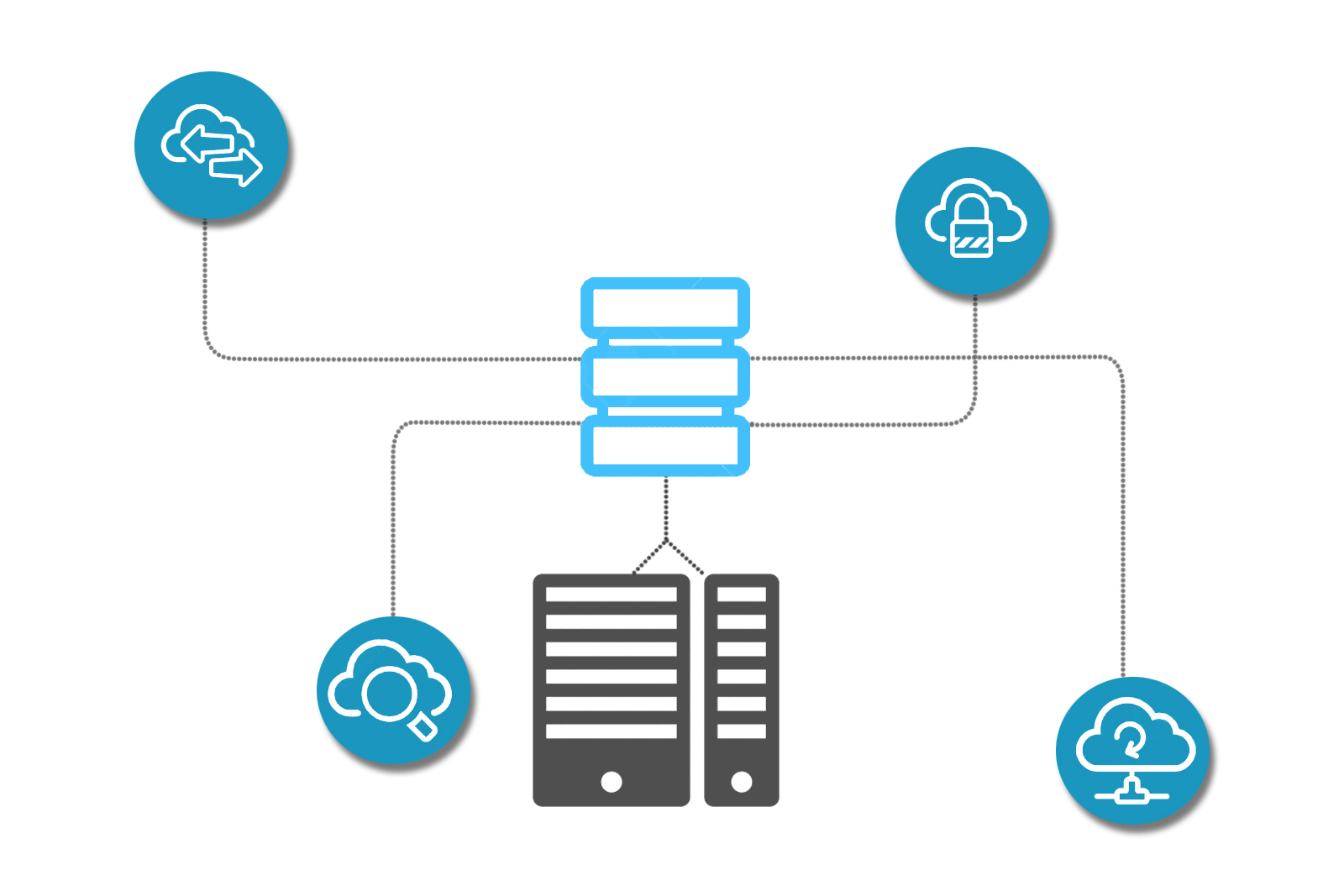
Centralized Imaging Database
Our centralized imaging database helps authorized users easily find, view, download and share historical information regarding any phase of the study. Creating efficiencies by tracking the study progress and monitor the status of your case in our workflow. Whether it's digitized images, text-searchable documents or reports and read history – proprietary platform CLINSpa makes your files accessible to your global audiences. Multicentric trials are hereby made possible by acquisition of images across multisite globally. US government approved and HIPAA compliant archival system protects all DICOM images and the reports from natural disaster.
Get In Touch
Integrated Image management Solutions for every phase of your clinical trial that will optimize your costs & offer best in class Image Core Labs solutions. Be it in any Phase (Phase I. Phase II, Phase III or Phase IV) of clinical trial of pharmaceutical, biotechnology or medical device product, we are one stop solution for all your imaging needs.

Email Us: info@imagecorelab.com
Call Us:+91 8077198066
Give us a call or drop a message, we endeavour to answer all your queries within 24 hours on business days.

