
Clinical trials are an important part of the drug development process. They are essential in assessing the effectiveness of a new therapy as well as comparing the safety and efficacy of a new molecule with an existing one. Unfortunately owing to structural, infrastructural and procedural barriers, the failure rates of clinical trials are quite high. The cost of performing a clinical trial is exorbitant and is considered as one of the leading cause of the decline in the number of new FDA submissions. Developing new methods and techniques to reduce the effort and cost of a clinical trial is the need of the hour.
Common challenges in Clinical Trials
- Various on-ground challenges
- Challenges in recruiting the patient as per the inclusion and exclusion criteria
- Data collection challenges
- Training of the site personnel
- Lack of supportive infrastructure
Imaging in Clinical Trials
In March of 2004, the FDA announced plans to reform the regulatory path to reduce the barriers to new drug development. Imaging was identified as one of the components that could play an important role in reducing the cost of clinical trials. Previously imaging was particularly used in phase 1 and phase 2 trials of therapeutic drug development, but of late imaging studies have been proposed for phase 3 trials as a component for the primary and secondary endpoints. Over the last few decades imaging has become increasingly important in determining the endpoints such as time to progression or progression-free survival. From an imaging perspective, tumour progression is defined as either an increase in existing tumour size or detection of newer lesions. In the era of cytotoxic therapy, the response rate (the proportion of a tumour that shrank) was the standard endpoint of the oncological trials which was done through imaging.
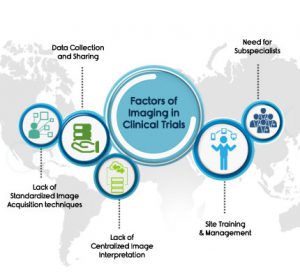 Factors that limit the role of Imaging in Clinical Trials
Factors that limit the role of Imaging in Clinical Trials
Clinical trials are incredibly complex, and complexity increases when it comes to multicentric trials with the huge enrollment of patients. Regardless of the therapeutic area, the phase of trial or indication, adding imaging as a part of a trial further complicates it by new regulatory and workflow compliance challenges.
Below is a list of a few of those challenges:
Site Training & Site Management: Adding imaging in a clinical trial requires training of the site staff on various imaging requirements. A successful image interpretation is not just limited to equipping sites with proper tools and software; it also requires training of technologists for the appropriate imaging protocol. For example, a majority of the errors produced in image capture include technical errors such as motion artifact, blurred image, pressure mark, incorrect positioning; exposure errors ; processing and post processing errors ; contrast dose timing errors and incorrect slice selection in CT etc. Capturing the right images is crucial for a successful image interpretation, and a successful clinical trial. Defining the correct imaging parameters in the study protocol is therefore of great importance.
Need for Subspecialists: A given trial can have a variety of modalities that require review by clinical expert readers. For examples in oncology clinical trials, therapy is assessed using imaging response criteria involving imaging biomarkers. Various Imaging response criteria are used to determine the endpoints. Imaging biomarkers are characteristics extracted from images under study and may vary with a tumour or cancer types and with imaging modalities. Response criteria are created using specific imaging biomarkers with metrics and sets of rules, to provide accurate and reproducible measurements of tumours during therapy.
Data Collection and Sharing: The lack of mechanisms for easy sharing imaging data across multicentric minimizes the use of imaging in clinical trials. Access to a cloud based teleradiology workflow platform which has anonymization tools as well as dual read and adjudication workflow features can optimize the conduct of the image interpretation of a clinical trial.
Lack of Standardized Image Acquisition techniques: Standardization of Imaging Acquisition technique in medical practice differs from a clinical trial. In case of clinical trials, standardization is confined to the protocol requirement, which defines the nature of the imaging to be performed and the frequency of imaging evaluation. For instance, a trial protocol may require all subjects to undergo imaging both with and without contrast enhancement and so on.
Standardization involves determining whether the specification and the requirements of the protocol (with regards to imaging aspect) are correct regarding the number of images to be captured, timing of imaging during the trial, subject sedation, positioning, image interpretation and display as well as image archival. This is particularly important in multi-geographic trials where there are different technicians in different locations. The need for adequate training is imperative as a deviation from imaging standards will result in bias.
Every trial has its specific design and objectives; hence no single set of imaging standard will be readily applicable to all trial. Correlating and development of the right imaging protocol thereby defining the correct acquisition technique requires vast experience (on the ailment and other associated factors such as disease progression, number and dosage, timing etc.) of the radiologist. For example, an oncology trial imaging requires the knowledge of growth and pattern of a tumour; minimum drug dosage that will reflect the effect on the tumour making it visible in imaging.
A sponsor should consider the various component of imaging process standards when augmenting medical practice imaging process. Consistent image acquisition, processing and evaluation processes are not only important for ensuring imaging endpoint data quality, objectivity and reproducibility, they are also crucial for the seamless acquisition of images in a multicentric trial, avoid bias and in meeting regulatory standards for obtaining marketing approval.
Lack of Centralized Image Interpretation: The need for centralised image interpretation is contingent upon the role of imaging within the trial. A centralised image interpretation is critical to control bias in an open-label trial. A centralised imaging database helps authorised users easily find, view, download and share historical information regarding any phase of the study. An efficient database helps in tracking the study progress and the status of the case in the workflow. Furthermore, a centralised image interpretation allows for faster report reads ensuring right radiologist reads the right report. A centralised imaging database allows for easy access to all reports and reads history.
 How Image Core Lab Helps
How Image Core Lab Helps
At Image Core Lab we understand the unique challenges that pharmaceutical companies face when it comes to adding imaging in clinical trials. We provide all services necessary to support the Clinical Imaging Management including training of site personnel, equipping the site with proper tools, quality assurance of the captured images, quality check of the acquired images, expert reading, adjudication, formatted reporting, till the submission of EDC. Our cloud-based server protects all medical imaging records from any natural disaster and facilitates faster and easy retrieval.
Pool of experts
We bring you the same outstanding consistency and professionalism, backed by the pioneers of cross-border reporting- Teleradiology Solution. Our large team of expert advisory board and highly qualified, fellowship trained, and board-certified Radiologists provide an innovative environment for a broad range of image interpretation Scenarios.
Our therapeutic expertise
Our therapeutic expertise includes Oncology, musculoskeletal, central nervous system, neuro-oncology, neuro radiology, cardiac and vascular, thoracic and pulmonary imaging.
Oncology: Oncology trial is currently ICL’s fastest growing therapeutic area with over 50% of engagements in oncology trials. Experienced in large cancer trials with irRC and RECIST 1.1 protocols. Recently, RECIST and RECIST 1.1 were done together in the same trial.
Neurological: Strategic and practical support from experts that will boost your trial process and simplify your product development process. Our fellowship-trained neuroradiologists bring in a subspecialty level experience on neurodegenerative diseases such as Alzheimer’s Disease and Parkinson’s Disease, demyelinating disorders, stroke, epilepsy, neuro-oncology and post-traumatic disorders.
Cardiac and vascular: Our strength in Cardiac MR (CMR) image analysis, includes Anatomic Perfusion, Functional Imaging, Graft Patency and Stent Studies, as well as expertise in Nuclear Cardiology, further complements these services. A global team of in-house cardiovascular experts, who are highly-experienced in image analysis of clinical research and drug development.
Thoracic and pulmonary imaging: We provide the highest level of expertise in the evaluation of chest radiographs and high-resolution CT of pulmonary disorders, including interstitial lung disease, chronic obstructive pulmonary disease, occupational lung diseases, pulmonary thromboembolism, bronchopulmonary infections and tumours.
CLINSpa- technology
A web-based platform that’s convenient and customizable designed tried and tested for an end to end management of imaging trial workflow. It allows you to customize workflows output templates thereby automating the process.
We aim to leverage our deep set expertise in technology and image interpretation service to accelerate drug development trials at the right speed.
To find out more about our Services, or how we can help you manage imaging requirements of your trial talk to us today.



0 Comments